Sickle cell disease (SCD) is an inherited red blood cell disorder caused by a point mutation in hemoglobin formation that affects millions of people across the world. In sub-Saharan African countries, the prevalence of SCD reaches as high as 45%, and more than 300,000 children are born each year with SCD. Due to the lack of proper diagnosis and appropriate treatment, many of these of children will die due to SCD.
Different methods have been developed for screening and diagnosis of SCD. However, most of these methods require costly, benchtop devices, or are based on human reading. Human errors along with storage requirements, e.g. controlled temperature and humidity to preserve chemical activity/function, limit the effectiveness of these tests, including various point-of-care (POC) assays.
Microscopic examination of peripheral blood smear slides for sickle cells has been used as an alternative method to diagnose SCD for many years. Preparation of these blood smear slides can be easily performed in sub-Saharan Africa as there are hundreds of thousands of these slides prepared each year to diagnose other parasitic infections and blood disorders. However, microscopic inspection of blood smear slides for SCD diagnosis requires an expert to make the diagnosis and can be prone to errors due to lack of sufficient training and quality control, especially in resource limited settings.
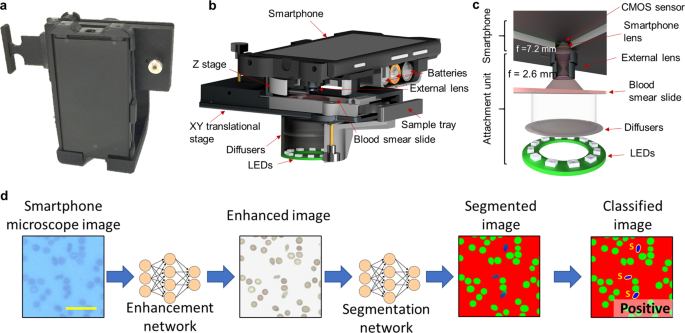
The strong need for improved access to testing led us to develop an automated method to diagnose SCD using images of a blood smear captured using a cost-effective mobile microscope [1]. As smartphones already have high-quality cameras, efficient processors, and connectivity to enable remote monitoring, we transformed a smartphone into a mobile microscope and used it for imaging blood smears. This smartphone-based microscope (see Fig. 1) uses an attachment which connects optical lenses and light sources to the rear-facing camera of the phone and enables sub-micron resolution imaging of microscope slides.
In order to eliminate the need for a trained expert to analyze the acquired images, a deep learning-based framework (see Fig. 2) was developed to automatically perform the SCD analysis. This framework uses two complementary deep neural networks, one feeding the other. The first network is used to enhance and standardize the blood smear images captured by the smartphone microscope. The second network acts on the output of the first image enhancement network and performs the segmentation between healthy and sickle cells within the acquired image, which is then used to rapidly determine the diagnosis per patient. This mobile sickle cell detection method was blindly tested using blood smears from 96 unique patients, including 32 SCD patients, and we achieved ~98% diagnostic accuracy, with an area-under-the-curve (AUC) of 0.998.
Our results reveal that this method is highly accurate, efficient, and cost effective, taking only a few seconds to perform the analysis on a remote or local computer, after the images are acquired using the mobile microscope. Looking forward, we believe that this technology has the potential to improve access to testing for sickle cell disease and other blood cell disorders in resource limited areas, where current testing techniques are insufficient.
[1] K. de Haan, H.C. Koydemir, Y. Rivenson, D. Tseng, E. Van Dyne, L.S. Bakic, D. Karinca, K. Liang, M. Ilango, E. Gumustekin, and A. Ozcan, “Automated screening of sickle cells using a smartphone-based microscope and deep learning,” npj Digital Medicine DOI: 10.1038/s41746-020-0282-y (2020)

Figure 1: (a) Photograph of the smartphone-based microscope. (b) Design schematics of the portable microscope. The microscope is built as a 3D printed external attachment, allowing the smartphone to capture sub-micron resolution images of blood smears.

Figure 2: Blindly tested image patches demonstrating the automated workflow used to classify the imaged blood smears. Each acquired smartphone image first passes through an image enhancement and standardization network. A second, separate network is then used to segment these enhanced images and determine the number of normal and sickled cells within each image. Based upon the number of normal and sickled cells counted, a diagnosis is made. Scale bar indicates 20 μm.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in